Solutions for hygienic and cost‑efficient oligonucleotide production
The production of oligonucleotides requires systems that can safely handle toxic and flammable chemicals while meeting strict GMP and hygienic‑design standards. At the same time, equipment must offer flexible scalability—from pilot‑scale setups to full industrial manufacturing.
Precision dosing and mixing for upstream and downstream processing
Diaphragm metering pumps enable accurate dosing of reactive synthesis fluids and contamination‑free downstream processing. A key requirement is the ability to prepare toxic or flammable mixtures flexibly, even when flow rates vary significantly.
High‑precision dosing of reactive chemicals
Stable mixing performance across wide flow‑rate ranges
Suitable for both synthesis and purification steps
Hygienic metering technology: LEWA ecodos hygienic
For GMP‑compliant downstream processes, LEWA ecodos hygienic pumps are used. They offer:
- Corrosion resistant, hygienic design
- Four layer sandwich safety diaphragms
- Certified materials (FDA, USP, AOF)
- Electropolished surfaces (Ra ≤ 0.5 µm)
- Nearly dead space free construction for fast CIP cleaning
- EHEDG EL Class 1 certification
- Materials such as 1.4435, 1.4529, or Hastelloy for aggressive media
Inline dilution for consistent buffer quality
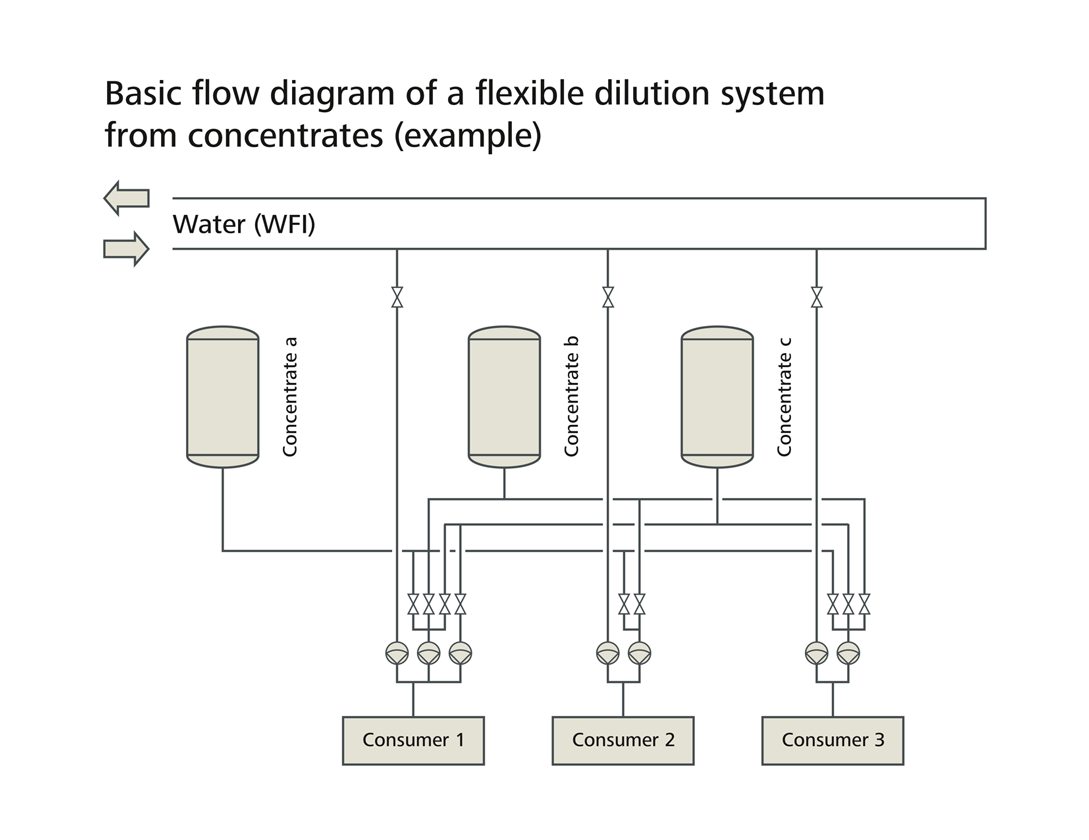
Stable chromatography performance depends on reproducible buffer composition. LEWA inline dilution systems provide:
- Precise, continuous dilution
- Multi‑channel configurations with valves, multi‑head diaphragm pumps, and Coriolis flow meters
- Low‑pulsation mixing via dynamic pressure control
- Prevention of pressure fluctuations, gas entrainment, and heat input
- Integrated pH and conductivity monitoring
- Centralized evaluation of all sensor data at high sampling rates
This ensures buffer solutions are always available in the required quality for semi‑continuous chromatography.
Custom package units for downstream processing

LEWA develops tailor‑made inline metering and dilution systems:
- Delivered as complete package units (PU)
- Compatible with solvents such as ethanol, isopropanol, toluene, and acetonitrile
- Designed for Ex Zone 2 IIB T3
- Flow ranges from a few l/h up to 6,000 l/h
- Multi head diaphragm pumps for low pulsation flow
- Back pressure independent characteristics for reproducible continuous dosing
These solutions enable safe, scalable, and cost efficient oligonucleotide production—from synthesis to final purification.
Background
Growing importance of therapeutic oligonucleotides
The demand for oligonucleotide‑based therapeutics is rising steadily — both for rare genetic disorders and for widespread chronic diseases. Oligonucleotides, short RNA or DNA sequences, are among the most essential tools in modern molecular biology. They are composed of nucleotides, the fundamental building blocks of nucleic acids, and can be precisely modified through industrial DNA and RNA synthesis to achieve targeted therapeutic effects.
Therapeutic approach: targeting genetic dysfunction
In contrast to conventional biopharmaceutical agents that primarily target proteins, therapeutic oligonucleotides act directly on defective genetic mechanisms. This direct mode of action makes them highly promising for previously untreatable diseases, such as specific neurodegenerative disorders and rare inherited conditions. Through precise interaction with RNA, oligonucleotides can modify gene expression, correct aberrant splicing, or inhibit harmful transcripts, offering a powerful platform for next‑generation genetic medicines.
A milestone in this field was reached in 1998 with the approval of Fomivirsen (Vitravene), the first antisense oligonucleotide used to treat CMV infections in patients with AIDS1. Subsequent approvals followed, including Patisiran (Onpattro) in 2018, a lipid‑nanoparticle‑based therapy for polyneuropathy caused by hereditary transthyretin amyloidosis. These developments underscore the growing clinical relevance of antisense therapies, RNA interference (RNAi), and other oligonucleotide‑based modalities in modern medicine.
Industrialization: Transitioning oligonucleotide therapies from lab scale to GMP production
Although several oligonucleotide‑based drugs have already been approved, the industrial‑scale manufacturing of many of these therapies is still under development. With numerous candidates progressing through clinical trials, the focus is increasingly shifting toward process optimization, scalability, and regulatory compliance. To meet rising demand, pharmaceutical manufacturers must establish GMP‑compliant production systems that can reliably scale from laboratory and pilot operations to full industrial output — without compromising yield, purity, or overall efficiency. This makes scalable oligonucleotide production a critical success factor for the next generation of RNA‑ and DNA‑based therapeutics.
¹ Text information from the ABDATA pharmacy database.
Industries
Pompes et systèmes pour les industries pharmaceutique, des sciences de la vie et biotechnologique
En tant que fabricant leader de pompes et systèmes de pompes pour les industries pharmaceutique, des sciences de la vie et biotechnologique, LEWA vous aide à mettre au point des solutions complètes, parfaitement adaptées à vos procédés. Lors de la fabrication de produits pharmaceutiques, un savoir-faire particulier est de mise pour pouvoir assurer le respect des prescriptions légales. La fiabilité de la production, la documentation et l’aptitude à la qualification sont primordiales à nos yeux.
More DetailsProducts
Pompe doseuse à membrane LEWA ecodos® sanitary/hygienic
Une version sanitary/hygienic est également disponible pour LEWA ecodos. Elle satisfait à toutes les exigences d'utilisation dans la production pharmaceutique et alimentaire.
More DetailsSystèmes de dosage personnalisés LEWA
Nous proposons des systèmes de dosage intégrables directement dans un circuit de régulation avec surveillance et contrôle de la cohérence des données. Vous bénéficiez ainsi d’un système de dosage unique, doté d’une pompe optimale pour le dosage de fluides dans le cadre de processus automatisés.
More Details